Coma/Near Coma Scale (CNC)
A bedside tool for detecting subtle change in patients with the most severe disorders of consciousness
(Rappaport et al., 1992)
The Coma/Near-Coma Scale (CNC) is a standardized neurobehavioral assessment developed to measure small clinical changes in adults with severe brain injuries who function at very low levels of consciousness (Rappaport et al., 1992). Patients who would otherwise plateau on broader scales such as the Glasgow Coma Scale or the Disability Rating Scale.
Where the GCS asks “is the patient in coma?” and the CRS-R asks “what level of consciousness is emerging?”, the CNC zooms in on the bottom of the curve. It was designed to give clinicians a way to document the kind of incremental, day-to-day movement that families and bedside staff so often notice but that broader instruments miss.
Target Population
Adults with severe traumatic or non-traumatic brain injury.
Patients in coma, the vegetative state / unresponsive wakefulness syndrome, or the low end of the minimally conscious state.
Anyone whose responses are too inconsistent or too subtle to be reliably captured by the GCS, FOUR score, or full CRS-R.
The CNC is most useful when the clinical question is “Is this patient changing at all, in either direction?” rather than “What is their diagnosis?”
The 11 Items
Each item delivers a standardized sensory or motor stimulus and asks the examiner to rate the most meaningful response observed across repeated trials.
Auditory: Response to bell ringing presented at midline.
Command: Response to a verbal command (e.g., “open your eyes,” “squeeze my hand”).
Appendage: Response to passive limb movement (e.g., flexion/extension of an arm).
Visual: Eye-opening and visual orientation toward a stimulus presented in the visual field.
Threat: Reaction (e.g., blink, withdrawal) to a visual threat (rapid hand approach toward the eyes).
Olfactory: Response to a noxious odor (e.g., ammonia capsule held under the nose).
Tactile (shoulder): Response to a firm tap or squeeze on the shoulder.
Tactile (nasal swab): Response to a cotton swab gently inserted into the nostril.
Pain: Response to a noxious stimulus (e.g., nail-bed pressure).
Vocalization: Spontaneous or stimulus-elicited vocalization (sounds, words).
Eye position at rest: Resting gaze and eye position observed before stimulation.
Note: Some published versions of the CNC report 8–10 scored items because pain and certain tactile items are sometimes omitted in research protocols (e.g., pain-free indices). Make sure your unit specifies which items are in use and document accordingly.
Scoring
Each of the 11 items is rated on a 3-point scale of 0, 2, or 4. Lower scores indicate better function — the opposite direction from the GCS and CRS-R, which is a common source of charting errors and worth flagging in your handoff.

Calculating the Level
Sum the item scores
Divide by the number of items administered to get the mean score
Match the mean score to one of the five levels below
The Five Levels
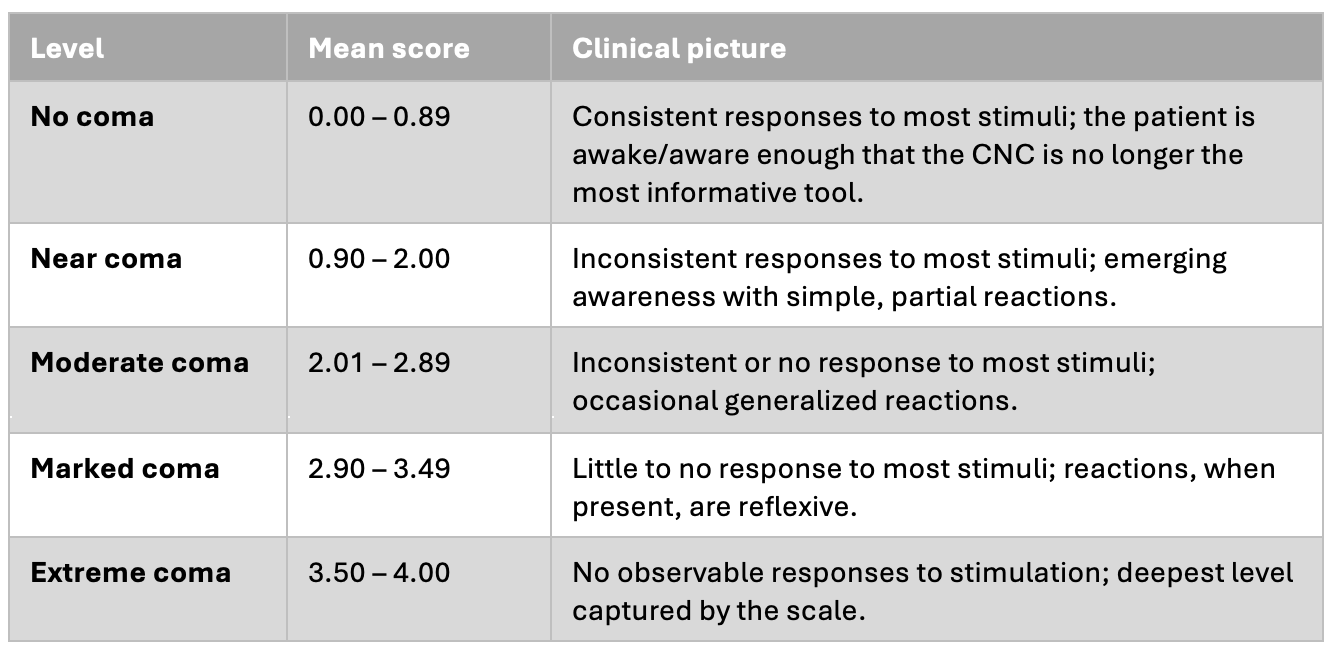
Direction matters. A falling mean score is improvement; a rising mean score is decline. Document the trend, not just the snapshot.
How to Administer
Setup
Quiet room; minimize visitors, alarms, and overhead pages for the duration of the exam.
Patient positioned with head of bed elevated 30–45° if tolerated.
Stimuli on hand: bell, penlight, ammonia capsule (or other noxious odor), cotton swab, gloves.
Eyes uncovered; suction the airway and clear secretions before starting if needed.
During the exam
Present each stimulus at least three times, with brief pauses between trials.
Score the best, most consistent response across trials — not the first or the last.
Pause if the patient becomes physiologically unstable (HR, BP, ICP, SpO₂) and resume when safe.
Frequency
For change detection, the CNC is typically administered twice daily for three days, then re-run at clinically meaningful intervals.
Always note time of day, time since last sedation/analgesia, and any acute medical events — each of these can move the score.
How well does it perform?
Original validation (Rappaport et al., 1992) reported interrater reliability of r = 0.95 in low-level patients.
Validity supported by significant correlations with multimodality evoked-potential abnormality scores and with the Disability Rating Scale.
Subsequent Rasch analysis (Hauger et al., 2020) found Person Separation Reliability of 0.87–0.89, supporting use for group-level decisions in adults with disorders of consciousness.
Sensitive to small changes that broader scales miss — particularly useful for tracking response to medications, weaning sedation, or recovering after a medical setback.
Strengths and limitations
Strengths
Quick to learn and inexpensive to run; the bedside kit fits in a pocket.
Designed for the population that other consciousness scales handle worst.
Repeated administration creates a defensible, longitudinal record of response.
Aligns naturally with family-facing conversations: each item is concrete and observable.
Limitations
Less granular at the upper end — once the patient is consistently following commands, transition to the CRS-R or another tool.
Reverse scoring (low = good) is unintuitive and easy to misread; consider a chart legend.
Confounders matter: sedation, analgesia, paralytics, fever, intracranial hypertension, electrolyte derangements, and recent seizures can all suppress responses.
Pain items are ethically contested in some settings; check your unit’s policy before including them and document any modifications.
Putting it into practice
If your patient has been in a coma for more than 72 hours and you are no longer seeing meaningful change on the GCS, try adding a CNC to the morning and evening neuro check. Pair the score with a one-line narrative: what stimulus produced the most informative response, and what that response looked like. Over a week, this becomes one of the most useful documents in the chart — for the family, for the consult team, and for whoever is making the next major decision about goals of care.
References
Rappaport, M., Dougherty, A. M., & Kelting, D. L. (1992). Evaluation of coma and vegetative states. Archives of Physical Medicine and Rehabilitation, 73(7), 628–634.
Rappaport, M. (2000). The Coma/Near Coma Scale. The Center for Outcome Measurement in Brain Injury. http://www.tbims.org/
combi/cnc
Rappaport, M. (2005). The disability rating and Coma/Near-Coma scales in evaluating severe head injury. Neuropsychological Rehabilitation, 15(3–4), 442–453. https://doi.org/10.1080/09602010443000335
Hauger, S. L., Løvstad, M., Andelic, N., Sigurdøttir, S., Schanke, A.-K., Røe, C., & Søberg, H. L. (2020). Psychometric properties of the Coma Near-Coma Scale for adults in disordered states of consciousness: A Rasch analysis. Archives of Physical Medicine and Rehabilitation, 101(11), 1965–1974.
Weaver, J. A., et al. (2023). Comparing indices of responsiveness for the Coma Near‐Coma Scale with and without pain items: An exploratory study. Brain and Behavior, 13(7), e3120.
Traumatic Brain Injury Model Systems. The Coma/Near Coma Scale (CNC scoring form, tbims.org).

